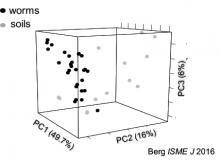
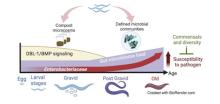
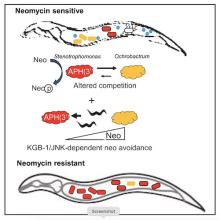
Research in our lab seeks to understand the fundamentals of host-microbe interactions in the context of the whole organism, using the powerful genetic model Caenorhabditis elegans as a model host. We focus on the role of host genetic factors in shaping gut microbiome structure and function, how gut commensals affect host immunity and host-pathogen interactions, and how aging changes all that (with special interest in the role of antagonistic pletriopy in aging). Our level of investigation spans the range between molecular mechanisms, ecological constraints and impact on evolution.
Looking for a place for your Ph.D? Join us!