Laboratory Hypotheses
The Lactate Shuttle: The
Shuttling of lactate between cell compartments, cells, tissues and organs is a
major means of distributing carbohydrate energy, supporting glycemia via
gluconeogenesis, and intracellular as well as cell-cell signaling. Lactate is formed continuously, but
especially after carbohydrate nutrition and during physical exercise. Most (70-80%) of lactate formed during
exercise is oxidized and used as a fuel energy source. Working red skeletal
muscle and heart are fueled by lactate during exercise. Other tissues, such as brain, also use
lactate as a fuel during exercise. The remainder of lactate disposal (20-30%)
during exercise is for conversion to glucose in the liver and kidneys, but the
magnitude of lactate turnover during exercise is so great that lactate is the
main gluconeogenic precursor. As
well, in it’s role as a signaling molecule, lactate has been characterized as a
“Lactormone.” In our laboratory we study the
regulation of Cell-Cell
and Intracellular lactate Shuttles and of the cell protein constituents that facilitate functioning of
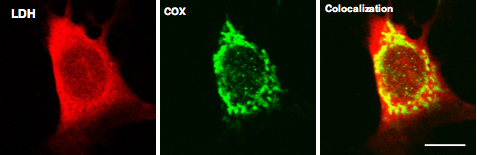
lactate shuttles. Micrographs at the Top of the page show organization of
components of the Mitochondrial Lactate Oxidation Complex in cultured mammalian
muscle fibers.
 The Crossover
Concept: Information on the relative use of fuel energy substrates
(carbohydrates, fats, proteins) has been used to develop a model of Energy-Substrate
Partitioning in which the
effects of exercise intensity, gender, endurance training and nutrition are
coordinated and regulated. The Crossover
Concept holds that during
post-absorptive resting conditions, in muscle and at the whole body in general,
fats are the major fuel sources.
But, as exercise intensity increases, in working muscle there occurs a
switch (Crossover”) from dependence on fats to carbohydrate energy forms as
fuel sources. In this context,
some amino acids, such as the essential amino acid leucine are used, but in
general most amino acids are not important muscle energy sources. A model of the Crossover Concept in which the effect of relative exercise intensity
is depicted is shown below.
The Crossover
Concept: Information on the relative use of fuel energy substrates
(carbohydrates, fats, proteins) has been used to develop a model of Energy-Substrate
Partitioning in which the
effects of exercise intensity, gender, endurance training and nutrition are
coordinated and regulated. The Crossover
Concept holds that during
post-absorptive resting conditions, in muscle and at the whole body in general,
fats are the major fuel sources.
But, as exercise intensity increases, in working muscle there occurs a
switch (Crossover”) from dependence on fats to carbohydrate energy forms as
fuel sources. In this context,
some amino acids, such as the essential amino acid leucine are used, but in
general most amino acids are not important muscle energy sources. A model of the Crossover Concept in which the effect of relative exercise intensity
is depicted is shown below.
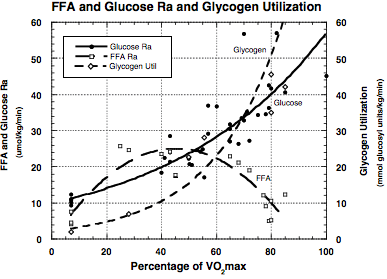
Figure: Results of
an extensive literature search showing blood glucose and free fatty acid flux
rates (Ra) and net muscle glycogenolysis as functions of relative exercise
intensity as given by % VO2max. This
form of analysis indicates exponential increments in muscle glycogenolysis and
glucose Ra's as functions of relative exercise intensity. In contrast, the analysis shows
multi-component polynomial response of plasma FFA flux, with easy to moderate
intensity exercise (i.e., 25-40% VO2max)
eliciting a large rise in flux, but crossover and decreasing flux at
approximately 55% VO2max. Plasma FFA flux is predicted to reach
minimal values as VO2max is
approached.
Recent Papers on the Lactate Shuttle
Brooks,
G.A, H. Dubouchaud, M. Brown, J. P. Sicurello, and
C.E. Butz. Role of mitochondrial lactate dehydrogenase and lactate
oxidation in the ‘intra-cellular lactate shuttle.’ Proc. Natl. Acad. Sci. USA
96: 1129-1134, 1999.
Brooks, G.A. Lactate shuttles in nature. Biochemical Society Transactions. 30: 258-264, 2002.
Hashimoto,
T., S. Masuda, S. Taguchi, and G. A. Brooks. Immunohistochemical analysis of
MCT1, MCT2 and MCT4 expression in rat plantaris muscle. J. Physiol. (London) 567: 121-129, 2005.
Hashimoto,
T., R. Hussien and G.A. Brooks. Colocalization of MCT1, CD147 and LDH in
mitochondrial inner membrane of L6 cells: Evidence of a mitochondrial lactate
oxidation complex. Am J Physiol Endocrinol Metab 290: 1237-1244, 2006.
Hashimoto,
T, R. Hussien, S. Oommen, K. Gohil, and G. A. Brooks. Lactate sensitive
transcription factor network in L6 myocytes: activation of MCT1 expression and
mitochondrial biogenesis. FASEB Journal Mar 29, 2007; [Epub ahead of print].
Recent Papers on the Crossover Concept
Kuo,
C.C., J. A. Fattor, G. C. Henderson, and G.A. Brooks. Effect of exercise
intensity on lipid oxidation in fit young adults during exercise recovery. J
Appl Physiol 99: 349-356, 2005.
Fattor,
J.A., B.F. Miller, K.A. Jacobs, and G.A. Brooks. Catecholamine response is attenuated during moderate
intensity exercise in response to the “lactate clamp.” Am J Physiol
(Endocrinol Metab) 288:
E143-E147, 2005.
Jacobs, K.A., R.M. Krauss, J. A. Fattor, M. A. Horning, A.L.
Friedlander, T. A. Bauer, T. A. Hagobian, E. E. Wolfel and G. A. Brooks.
Endurance training has little effect on active muscle fatty acid, lipoprotein,
or triglyceride net balances. Am J
Physiol Endocrinol Metab. 29: E656-665, 2006.
Friedlander, A.L., K. A. Jacobs, J. A. Fattor, M. A.
Horning, T. A. Hagobian, T. A. Bauer, E. E. Wolfel and G. A. Brooks.
Contributions of working muscle to whole body lipid metabolism vary with
exercise intensity and training. Am
J Physiol Endocrinol Metab. 292:
E107-E116, 2007
Wallis,
G.A., A.L. Friedlander, K.A. Jacobs, M.A. Horning, J.A. Fattor, E.E. Wolfel,
G.D. Lopaschuk, and G.A. Brooks. Augmented working leg glycerol turnover after
short-term endurance training in men. Am J Physiol Endocrinol Metab.
Response to Criticisms on Discovery of the Mitochondrial
Lactate Oxidation Complex: (https:// )